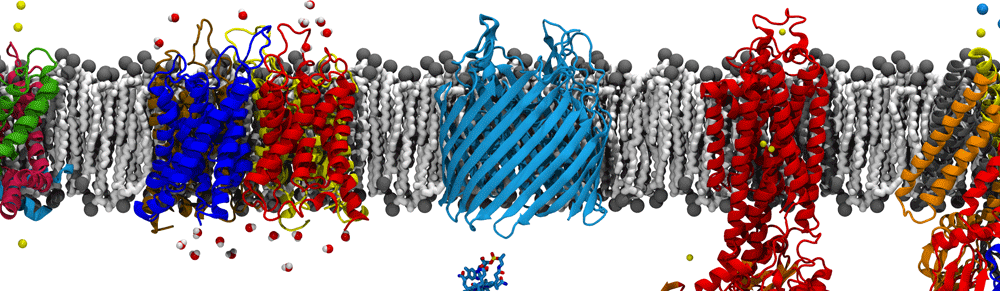
Every month, the Protein Data Bank highlights a specific molecule with a focus on its structure and its function. For the month of September, they are highlighting nanodiscs, which are tiny discs of lipids surrounded by two belt-like proteins. Originally engineered from high-density lipoproteins that mop up cholesterol in the blood, nanodiscs are most often used to study membrane proteins, which can fit inside, one per disc typically. Although widely used, the nanodisc structure is rarely seen. One of the first high-resolution images came from our 2011 paper on visualizing membrane-protein insertion through SecYE, embedded in a nanodisc, during translation by the ribosome, using cryo-electron microscopy and molecular dynamics simulations (see the image at right). Thanks to RCSB for bringing attention to this structure!
Author Archives: jcgumbart
Simulations and disease
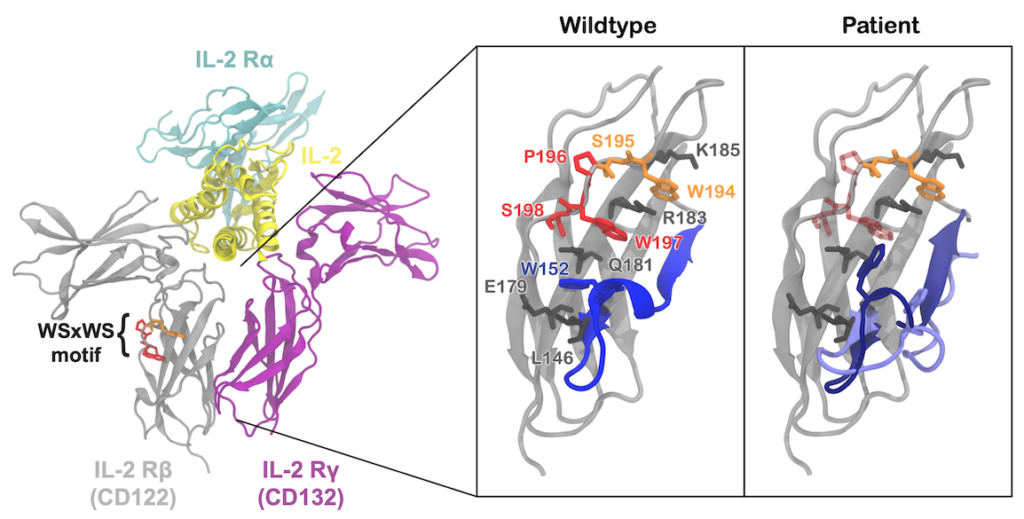
In most cases of disease, the cause is a complex interplay of various sources, including both genetic and environmental. But in some cases, the cause can be traced to a single mutation in a single gene. In two recent studies, molecular dynamics simulations contributed to our understanding of the function of the specific protein responsible for a particular disease. In the first study, simulations elucidated aspects of the signaling pathway in CFTR, a chloride channel that, when mutated, leads to the disease cystic fibrosis (study). In the second study, simulators teamed up with medical doctors to understand how a mutation to an IL2 receptor protein (pictured) may have led to severe immunological problems in a patient (study and news article). These two examples hopefully point the way towards bringing MD simulations closer to the bedside.
First graduates!
Congratulations to the first students to get their PhDs from the lab! Karl Lundquist, Anthony Hazel, Sunny Hwang, and Curtis Balusek all defended their PhDs at the end of last semester. Karl is sticking around for a couple months to finish up some projects before going to a postdoc with Nick Noinaj at Purdue, Anthony is already moving on to a postdoc with Alex MacKerell at U. Maryland, Sunny is going to do a postdoc in Emad Tajkhorshid’s lab at UIUC, and Curtis is starting a new career at Axis Group. We wish them all the best of luck!
Overcoming resistance
NAMD/VMD workshop in Dalian, China

JC and Anthony (along with Chris Chipot, UIUC and Edina Rosta, King’s College) just completed a 5-day NAMD/VMD workshop in Dalian China at the Dalian Institute of Chemical Physics, hosted by Guohui Li. In addition to all the great science discussed and tutorials completed, they also ate some great food and took in the sights. Hopefully this is the first of many there!